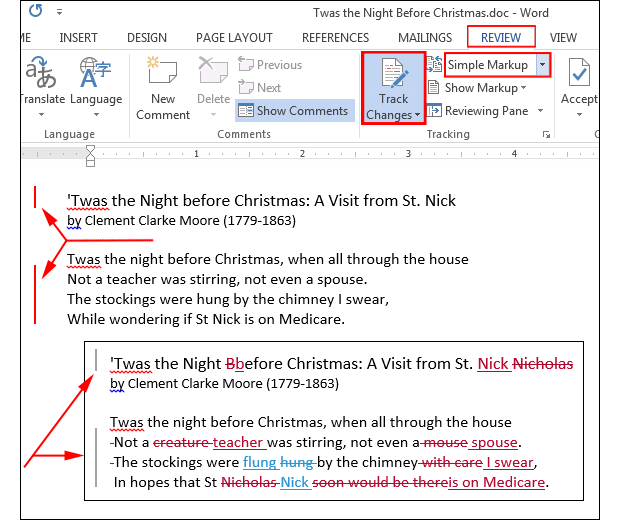
1 Induction of hepatocyte MAPK, p70S6k, and p90rsk by intraperitoneal injection of EGF. Mice were injected intraperitoneally with either saline or EGF. At given time points control and EGF-treated animals were sacrificed and livers were harvested. Cytosolic (panel A ) and nuclear (panel B ) extracts were prepared from isolated hepatocytes obtained by two-stage perfusion method (Protocol 1). Kinase activity was determined by immunocomplex kinase assays.

READY-TO-USE DEPOLYMERIZED 200mM SODIUM ORTHOVANADATE Prepared by pH10 adjustment-heating cycles With >99% vanadate stock high potency tyrosyl phosphatase inhibitor. Sodium Orthovanadate Activation Sodium orthovanadate should be activated for maximal inhibition of protein phosphotyrosyl-phosphatases. Prepare a 200 mM solution of.
Results are expressed as percentage kinase activation and represent means±S.D. Of 6 separate experiments. In these experiments basal cytosolic and nuclear MAPK activity averaged 4537±1630 and 1677±408, p70S6k activity averaged 15 899±4464 and 8753±15 400, and p90rsk activity averaged 16 815±2140 and 51 636±6324 c.p.m., respectively. A, b, and c indicate significant increase (a - p. 2 Induction of liver MAPK, p70S6k, and p90rsk by intraperitoneal injection of EGF. Livers were harvested at each time point from EGF- and saline-treated mice as described in Fig. Cytosolic (panel A ) and nuclear (panel B ) extracts were prepared from the liver after tissue pulverization (Protocol 2).
Kinase activity was determined by immunocomplex kinase assays. Results are expressed as percentage kinase activation and represent means±S.D. Of 4 separate experiments. In these experiments basal cytosolic and nuclear MAPK activity averaged 7107±2134 and 2972±434, p70S6k activity averaged 19 954±3026 and 10 383±1578, and p90rsk activity averaged 23 230±5176 and 64 208±10 239 c.p.m., respectively.
A, b, and c indicate significant increase (a - p. 3 Induction of hepatocyte MAPK, p70S6k, and p90rsk by intraperitoneal injection of sodium orthovanadate treatment. Cytosolic (panel A ) and nuclear (panel B ) extracts were prepared from isolated hepatocytes obtained by the two-stage perfusion method (Protocol 1) (Fig. Kinase activity was determinated by immunocomplex kinase assays. Results are expressed as percentage kinase activation and represent means±S.D. Of 6 separate experiments. In these experiments basal cytosolic and nuclear MAPK activity averaged 4070±1016 and 1506±367, p70S6k activity averaged 14 579±1366 and 7902±990, and p90rsk activity averaged 18 149±1482 and 49 500±11 995 c.p.m., respectively.

A, b, and c indicate significant increase (a - p. 4 Induction of liver MAPK, p70S6k, and p90rsk by orthovanadate treatment. Cytosolic (panel A ) and nuclear (panel B ) extracts were prepared from the liver after tissue pulverization (Protocol 2) (Fig. Kinase activity was determined by immunocomplex kinase assays. Results are expressed as percentage kinase activation and represent means±S.D. Of 4 separate experiments.

In these experiments basal cytosolic and nuclear MAPK activity averaged 7904±1672 and 3345±291, p70S6k activity averaged 17 594±4290 and 9384±1723, and p90rsk activity averaged 25 093±2741 and 60 733±8787 c.p.m., respectively. A, b, and c indicate significant increase (a - p. 7 Detection of cytoplasmic and nuclear MAPK, p70S6k, and p90rsk proteins and kinase activity.
The livers from control mice and those treated with EGF for 10 min were rapidly resected, frozen in liquid nitrogen, and after liver pulverization cytoplasmic and nuclear extracts were prepared. Equal amounts (4 mg) of cytoplasmic protein (Fig. 7A) or nuclear proteins (Fig. 7B) diluted in buffer A were separated by anion exchange chromatography using POROS HQ/M column, and the kinase activities were determined using immunocomplex assay (bottom graphs ) and kinase protein levels were determined by Western immunoblotting (top graphs ) protocols.

Column fractions were assayed as described in Materials and Methods. Similar elution patterns were obtained in 3 different experiments using different liver homogenates. Background/Aims: Although signal transduction pathways activated by EGF have been extensively studied in cultured cells, few such studies have been done in whole animals. In this study, activation of hepatic kinases, phosphatases, and DNA-binding activity of AP-1 was examined after intraperitoneal injections of either EGF or sodium orthovanadate into mice. Methods: Cytoplasmic and nuclear proteins, extracted from isolated hepatocytes or whole liver tissue, were immunoprecipitated with either anti-ERK1/2, anti-70S6k, or anti-p90rsk antibodies and kinase activities were measured using specific substrates.
Kinase protein levels was evaluated by Western blot analysis. AP-1 DNA binding activity was measured by electrophoretic mobility shift assay. Results: Systemic administration of EGF induced simultaneous increase in the activities of cytoplasmic and nuclear MAPK, p70S6k, and p90rsk.
MAPK and p70S6k were more potently activated in the cytosol while p90rsk activation was more pronounced in the nucleus. Orthovanadate also activated these kinases but to a much lesser degree than EGF. In vitro phosphatase assays showed that neither EGF nor orthovanadate induced measurable changes in phosphatase activities. EGF, but not orthovanadate, activated nuclear AP-1 DNA-binding activity in intact liver, indicating that activation of MAPK, p70S6k, and p90rsk by orthovanadate is not sufficient to activate this transcription factor. Conclusion: These observations provide groundwork for future studies to examine the role of EGF-induced kinase cascades and transcription factors in liver regeneration and other growth factor-mediated hepatic processes.
In transient forebrain ischemia, sodium orthovanadate as well as insulinlike growth factor-1 (IGF-1) rescued cells from delayed neuronal death in the hippocampal CA1 region. Adult Mongolian gerbils were subjected to 5-minute forebrain ischemia.
Immunoblotting analysis with anti–phospho-Akt/PKB (Akt) antibody showed that phosphorylation of Akt at serine-473 (Akt-Ser-473) in the CA1 region decreased immediately after reperfusion, and in turn transiently increased 6 hours after reperfusion. The decreased phosphorylation of Akt-Ser-473 was not observed in the CA3 region. The authors then tested effects of intraventricular injection of orthovanadate and IGF-1, which are known to activate Akt.
Treatment with orthovanadate or IGF-1 30 minutes before ischemia blocked delayed neuronal death in the CA1 region. The neuroprotective effects of orthovanadate and IGF-1 were associated with preventing decreased Akt-Ser-473 phosphorylation in the CA1 region observed immediately after reperfusion. Immunohistochemical studies with the anti–phospho-Akt-Ser-473 antibody also demonstrated that Akt was predominantly in the nucleus and was moderately activated in the cell bodies and dendrites of pyramidal neurons after orthovanadate treatment. The orthovanadate treatment also prevented the decrease in phosphorylation of mitogen-activated protein kinase (MAPK). Pretreatment with combined blockade of phosphatidylinositol 3-kinase and MAPK pathways totally abolished the orthovanadate-induced neuroprotective effect. These results suggest that the activation of both Akt and MAPK activities underlie the neuroprotective effects of orthovanadate on the delayed neuronal death in the CA1 region after transient forebrain ischemia.



























 RSS Feed
RSS Feed
