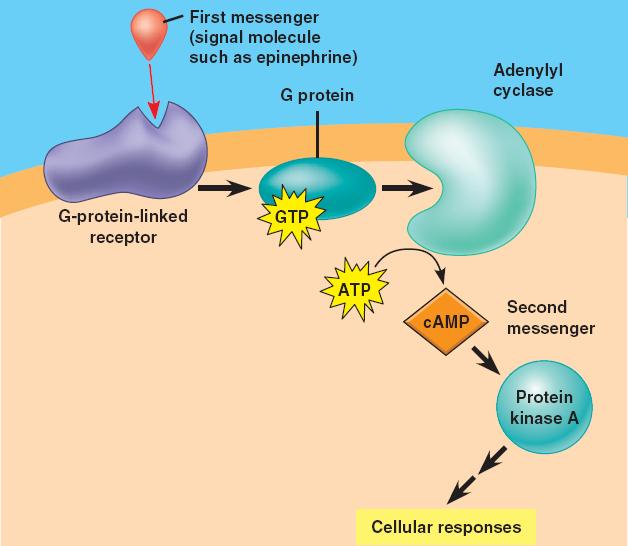
Forskolin is a potent adenylate cyclase activator, with binding (IC 50 =41 nM) to and activation (EC 50 =0.5 μM) of type I adenylyl cyclase. Intracellular cAMP signaling by soluble adenylyl cyclase. Neuronal cells, and. Inability to activate flagellar movement on ejaculation and. Furthermore, PKA and MAPK are coactivated in a subset of hippocampal CA1 pyramidal neurons following contextual fear conditioning. It is hypothesized that Ca2+ increases generated during formation of hippocampus-dependent memory activate CaM-stimulated adenylyl cyclases and that this cAMP signal supports the.
• 25 Downloads • Abstract Although both opioid receptors and endogenous opioids are abundant in cardiac tissues, the signal transduction pathways of opioids in cardiac sarcolemmal membranes have yet to be identified. In highly purified canine cardiac sarcolemmal membranes, binding of the opioid receptor antagonist [ 3H]diprenorphine and effects of μ, δ and κ agonists on low K m GTPase and adenylyl cyclase were measured. Equilibrium binding of [ 3H]diprenorphine revealed a maximal binding capacity of 7.2 pmol/mg protein and a K d of 1.3 nmol/1. In the presence of GTP, (D-Pen 2,5, p-Cl-Phe 4)enkephalin and (D-Arg 6)dynorphin A 1-13 fragment both inhibited adenylyl cyclase by 20–25% (from 206 ± 30to164 ± 28 pmolmin − 1mgprotein −1, EC 506 μmol/Landfrom254 ± 109to204 ± 90 pmolmin − 1mg protein −, EC 50 8 pmol/L, respectively; P.
The previous demonstration of an increase and redistribution of adenylate cyclase activity in injured peripheral nerve 1 suggests that an increase in neuronal cyclic AMP concentration could play a role in peripheral nerve regeneration. We report our finding that accumulating adenylate cyclase activity was translated into a twofold increase in cyclic AMP concentration in the regenerating nerve stump, coincident with the initiation and elongation of regenerative nerve sprouts. We sought to magnify the role of cyclic AMP in regeneration by using forskolin, a robust activator of adenylate cyclase 2, to produce an additional increase in neuronal cyclic AMP in situ.
Forskolin in vitro produced an approximately 40-fold greater elevation in neuronal cyclic AMP than an equimolar (10 −5) concentration of isoprenaline. Moreover, the elevated cyclic AMP concentration persisted for at least 60 min in the continued presence of forskolin. Daily injection of forskolin into the dorsal lymph sac of Rana pipiens, or delivery of forskolin through an implanted osmotic pump produced a sustained 40% increase in the rate of sensory nerve regeneration in freeze-lesioned sciatic nerves. We conclude that an increase in cyclic AMP concentration and, presumably, the activation of appropriate protein kinases stimulates regenerative nerve growth following trauma. 232, 413–424 (1982).

Cyclic Nucleotide Res. 7, 201–224 (1981). & Breckenridge, B. 26, 385–395 (1971). 66, 556–576 (1979). 234, 11–25 (1982).
Pichichero, M., Beer, B. Science 182, 724–725 (1973).

Gershenbaum, M. Neuroscience 5, 1565–1580 (1980). 63, 108–119 (1979). McQuarrie, I.
G., Grafstein, B. & Gershon, M.

132, 443–453 (1977). • • • • • 10.
& Berenberg, R. 156, 213–225 (1978).
• • • • • 11. & Hammerschlag, R. 24, 711–718 (1975). 279, 9–18 (1983). • • • • • 13. Biology and Pathology of Nerve Growth 161–235 (Academic, New York, 1981). M., MacFarlane, R.
& Cavanagh, J. 29, 95–107 (1976).
• • • • • 15. J., Murphy, R. A., Pichichero, M. Science 175, 73–74 (1972). Biochemical Action of Hormones, Vol.
8, 167–208 (Academic, New York, 1981). McQuarrie, I. In Posttraumatic Peripheral Nerve Regeneration: Experimental Basis and Clinical Implications, (eds A.
Gorio et al.) 49–58 (Raven, New York, 1981).
 RSS Feed
RSS Feed
